Biotechnology company Seagen Inc. (SGEN) and headquartered in Tokyo Astellas Pharma Inc. on Friday announced positive results from a Phase 3 trial of its investigational bladder cancer drug Padcev in combination with Merck's (MRK) Keytruda. p>
Key takeaways
- Biotechnology company Seagen and its Japanese partner Astellas Pharma have reported positive results. results from a late-stage trial of their investigational bladder cancer drug combined with Merck's Keytruda.
- The drug, Padcev and Keytruda reached key endpoints in patients with metastatic urothelial cancer.
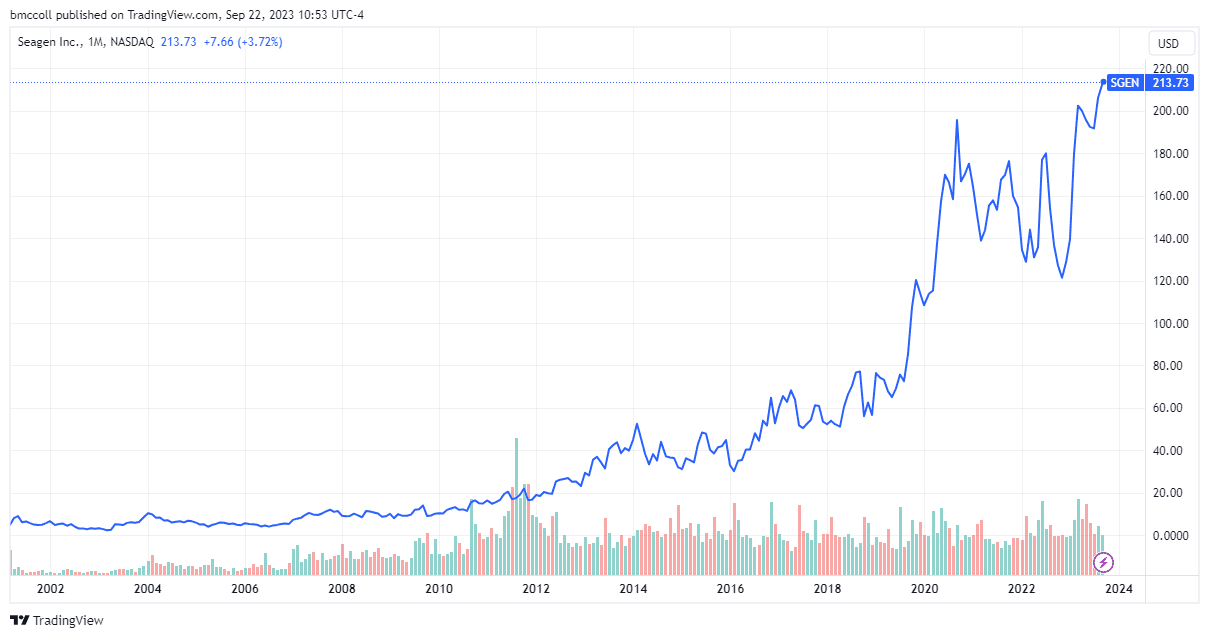
- After the news was reported on Friday, Seagen's shares reached a record level.
- Seagen and Astellas saidthe study aims to obtain accelerated approval of the drug combination from the Food and Drug Administration (FDA ) of the United States.
Padcev-Keytruda treatment was administered to those who had previously untreated locally advanced or metastatic urothelial cancer (la/mUC). , a form of bladder cancer that has spread to surrounding muscles, organs, or other parts of the body.
The companies said the study met key criteria for patients who received the drug combination versus those who took a placebo.
They noted that this study was intended to serve as the basis for global submissions and as a confirmatory trial for accelerated approval by the United States Food and Drug Administration (FDA). In April, the FDA gave accelerated approval to the Padcev-Keytruda treatment in certain adults with UC/mUC.
Dr. Ahsan Arozullah, senior vice president and head of oncology development at Astellas, said patients with “Metastatic urothelial cancer patients are in urgent need of additional treatment options and this combination has the potential to advance the standard of care. »
After When the news was announced, Seagen shares hit an all-time high. Merck shares were little changed midday Friday.
TradingView
Do you have a news tip for Investopedia journalists? Please email us at tips@investopedia.com
Source: investopedia.com