Takeaways
- Bristol -Stocks of Myers Squibb and 2770 bio fell after the FDA said it would not make a decision on expanded use of the company's stock. cancer drug next month.
- The treatment has already been approved by regulators to treat adults who have not responded to other drugs.
- A decision by Bayer ending its study of an experimental anticoagulant drug could also impact the future of a similar drug being tested by Bristol-Myers Squibb and Janssen Pharmaceuticals.
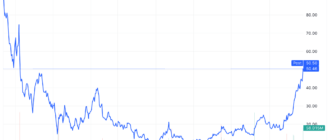
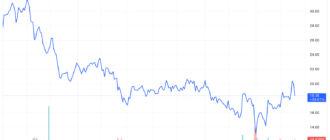
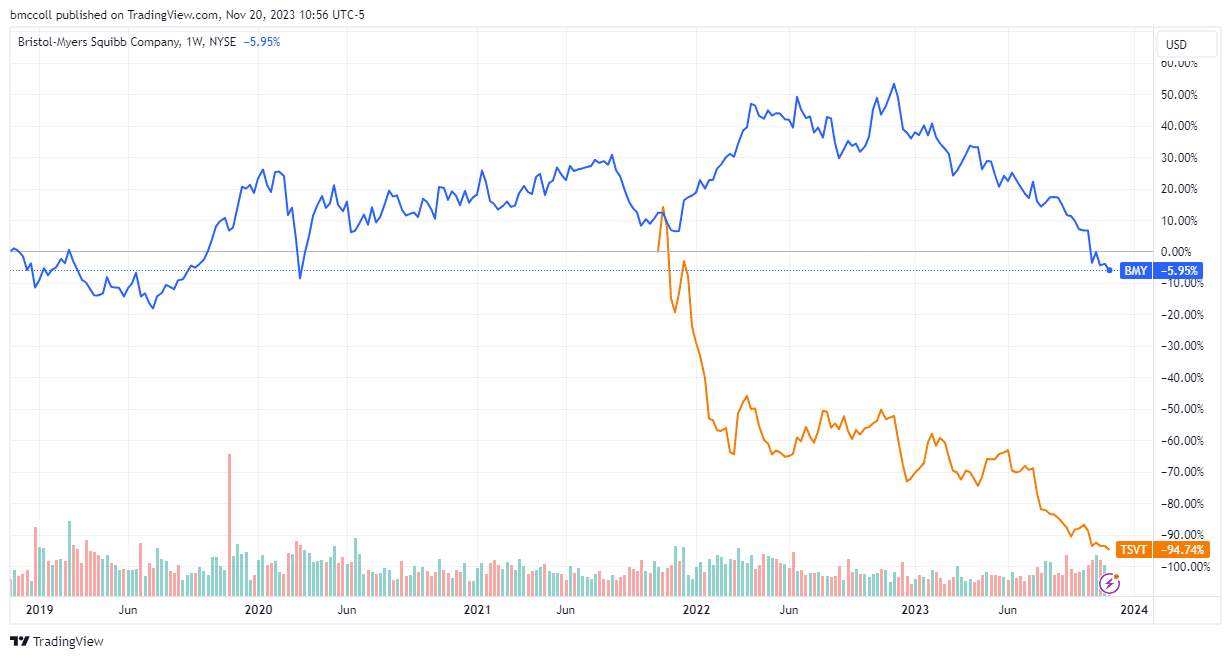
Bristol-Myers Squibb (BMY) Stock and its partner 2seventy bio (TSVT) fell after the two companies announced that the Food and Drug Administration (FDA) would not make a decision on expanded use of their blood cancer treatment, Abecma, next month , because initially planned.
The FDA was expected to decide Dec. 16 whether Abecma could be prescribed to people with prior lines of relapsed or refractory multiple myeloma exposed to three classes. Regulators had already approved Abecma to treat adult patients with multiple myeloma after four or more previous treatments proved ineffective.
Bristol-Myers Squibb and 2seventy bio said they anticipate officials will review data related to the secondary endpoint of overall survival rates in their Phase 3 study of Abecma. They added that they “look forward to continuing discussions with the FDA,” as well as participating in the FDA Oncology Drug Advisory Committee meeting that will analyze the results.
A decision by Germany's Bayer to end a phase 3 trial of an experimental anticoagulant drug due to a lack of effectiveness has also raised concerns about the #39;future of a similar drug being developed by Bristol-Myers Squibb and Johnson & Janssen Pharmaceuticals, a subsidiary of Johnson (JNJ).
Bristol-Myers shares Squibb fell more than 2% on Monday to their lowest level since the start of the COVID-19 pandemic, while 270 organic stocks fell more than 16% to their all-time low.

Trading View
Do you have any news tips for Investopedia journalists? Please email us at tips@investopedia.com
Source: investopedia.com